
The water cycle is the path that all water follows as it moves around Earth in different states. Liquid water is found in oceans, rivers, lakes, and underground. Solid water is found as snow or ice. The gaseous form of water is found as water vapor in Earth’s atmosphere.
To better understand the water cycle…
LET’S BREAK IT DOWN!
The Cycling of Water

Earth’s water is continuously being moved on, above, and below the surface of Earth. Water is always changing states between liquid, gas (vapor), and solid (ice). Water moves and cycles between the ocean, atmosphere, and land. Water evaporates from the surface of Earth, rises into the atmosphere, cools and condenses, and falls again to the surface as precipitation.
The water falling on land moves along the surface in rivers and streams and collects in bodies of water such as lakes and oceans. Most water that falls on the land soaks into the ground, where it can spend long periods of time trapped in the spaces between rocks and soil. Groundwater is the source of water for plants to grow, and humans have relied on groundwater as a drinking source for thousands of years.
Phase Changes

All matter is made of tiny moving particles called molecules. All matter can move from one state to another. The three normal states of matter are solid, liquid, and gas. However, some matter may require extreme temperatures and pressures in order to change states. Water, like all states of matter, requires the addition or removal of energy to change states. When heat energy is added to water, ice melts into a liquid and liquids evaporate into a gas. When heat is removed, gas condenses to a liquid and liquids crystallize into a solid.
Phase changes between the states of matter happen when you reach certain points of temperature and pressure. Water has a freezing and melting point of 0°C or 32°F. The freezing point is when liquid water turns to solid ice, and the melting point is when solid ice turns to liquid water. These are the same two transitions of states of matter between a solid and a liquid. Water has a boiling and condensation point of 100°C or 212°F. The boiling point is when liquid water turns into a gas, and the condensation point is when gas turns into a liquid. These are the two transitions of states of matter between liquid water and the gas form of water (called water vapor).
Sun Energy
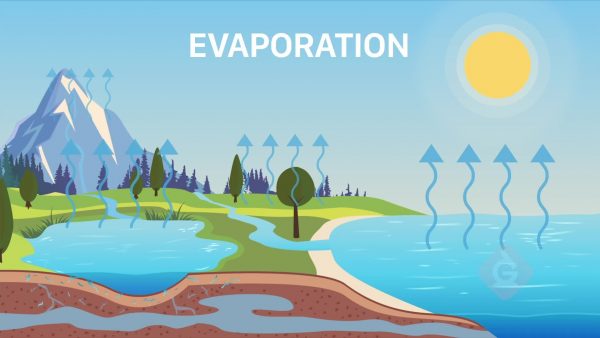
The solar radiation that comes from the Sun contains energy that drives the water to move through the water cycle. The Sun’s heat causes glaciers and snow to melt into liquid water. It also causes water to evaporate from oceans, lakes, and streams. The process of water evaporating from the leaves of plants is called transpiration. The Sun’s heat also warms water vapor, causing it to rise up through Earth’s atmosphere, where it condenses back into liquid water and creates clouds. All of these processes are driven by energy from the Sun and continually move water through the water cycle.
Gravity and Water Collection

Gravity is an attractive force that also helps move water through the water cycle. Water is pulled down from higher elevations by gravity into rivers, streams, and underground. Gravity also pulls water down from the atmosphere when it rains, snows, or hails. Most of the water that falls to the ground will soak into the ground and collect as underground water. When water reaches the ends of rivers or streams, it will begin to collect in bodies of water such as lakes or oceans.
Groundwater Flow

Water is continually moving underground downward and sideways because of gravity and pressure. Groundwater is a major contributor to flow in many streams and rivers and has a strong influence on river and wetland habitats for plants and animals. People have relied on groundwater for thousands of years as a source of drinking water and irrigation for agriculture.
The vast majority of groundwater occupies space between soil and rock particles. The ground acts like a sponge as it soaks up water into these spaces. At a certain depth below the land surface, larger spaces of collected groundwater can be found called aquifers. Water can also hit areas underground that are denser or have less space for water storage such as nonporous rock. This causes the water to flow in a more horizontal fashion, generally toward streams, oceans, or deeper underground.
WATER CYCLE VOCABULARY
WATER CYCLE DISCUSSION QUESTIONS
How does heat from the Sun turn liquid water into a gas called water vapor?
Explain how energy from the Sun drives the water cycle.
How do plants contribute to the water cycle?
How do clouds form?
How do snowflakes form?
How can the water molecules you drink today be millions of years old?
Skip, I will use a 3 day free trial
Enjoy your free 30 days trial



 GENERATION GENIUS
GENERATION GENIUS







